Osteoarthritis Pain Management
Monovisc: A High Molecular Weight Hyaluronic Acid Single Injection OA Treatment
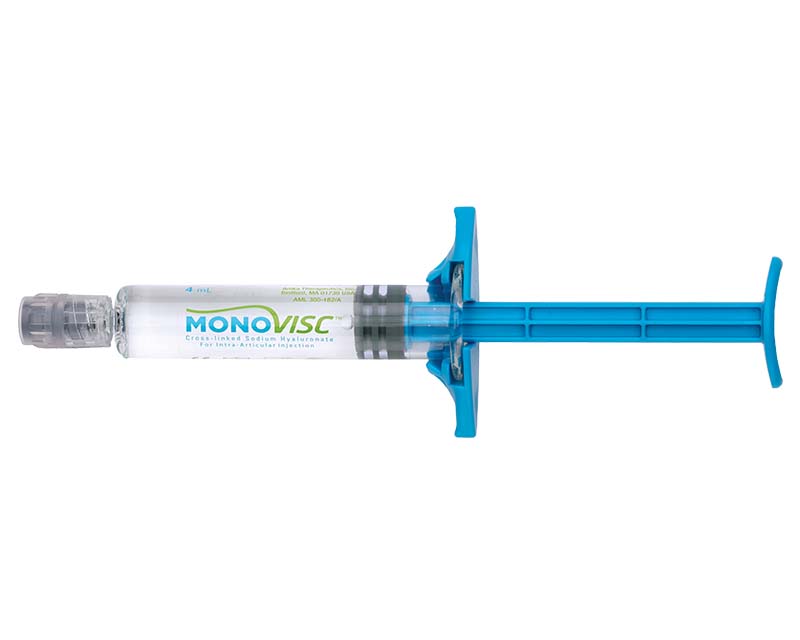
Monovisc® is a single injection viscosupplement utilized to treat knee joint pain caused by osteoarthritis (OA). It is comprised of highly purified, partially cross-linked sodium hyaluronate (NaHA) in a phosphate buffered saline solution, and it is both biocompatible and resorbable.
With just one injection, Monovisc may provide pain relief that lasts for up to 6 months.1 It is the only non-animal sourced hyaluronic acid (HA) single-injection treatment approved for use in the United States, and it delivers the maximum dose of HA (88mg) currently available in any single injection treatment.
Key Features & Benefits1,2,3:
- Convenient single injection treatment
- Highly concentrated, non-animal sourced HA
- Long lasting relief through 26 weeks
- Convenience of a single injection as opposed to multiple visits to the physician
- No confirmed pseudoseptic reactions reported
In the U.S., Monovisc is indicated for the treatment of pain caused by OA of the knee in patients who have failed to respond adequately to conservative non-pharmacologic therapy and to simple analgesics.
For complete product information, including indications, contraindications, warnings, and precautions, please refer to the Instructions for Use found here.
In Canada, Monovisc is indicated for the treatment of pain caused by OA of the knee in patients who have failed to respond adequately to conservative non-pharmacologic therapy and to simple analgesics.
In the European Economic Area, Monovisc is indicated as a viscoelastic supplement or a replacement for synovial fluid in human knee joints. Monovisc is well suited for treatment of the symptoms of human knee joint dysfunctions such as OA. The actions of Monovisc are lubrication and mechanical support.
For complete product information, including indications, contraindications, warnings, and precautions, please refer to the Instructions for Use provided with the product or Contact Us for a copy.
Anika products may not be available in all geographies. Product availability is subject to the regulatory clearances in individual markets. Please reach out to your local representative or Contact Us if you have questions about specific market approvals.
- MONOVISC 0702 Pivotal Clinical Trial. FDA Monovisc Summary of Safety and Effectiveness Data 2014
- Clinical Efficacy and Safety of MONOVISC. A lightly cross-linked highly concentrated hyaluronan specially formulated for single injection in osteoarthritis. White Paper Study conducted by Michael J. Daley, PhD. 2013
- Post-Approval Study of MONOVISC, A Symptomatic Treatment of Osteoarthritis, White Paper, 2012