Anika.
Restore Active Living.TM
Anika is a global joint preservation company committed to delivering meaningful advancements in early intervention orthopedic care, including osteoarthritis pain management, regenerative solutions, soft tissue repair, and bone preserving joint technologies.
Anika.
Restore Active Living.®
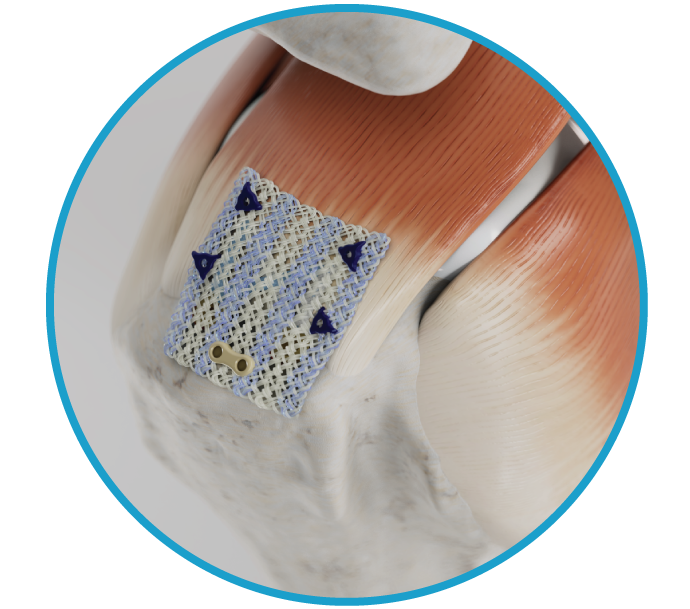
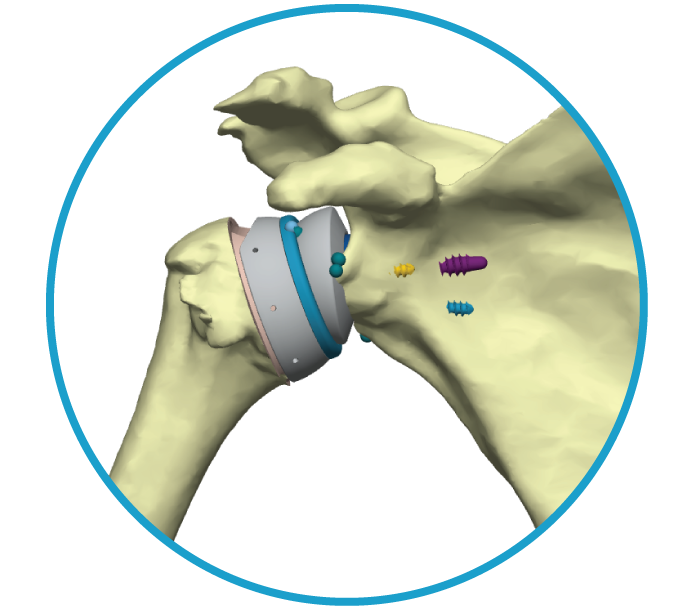
Anika is a global joint preservation company committed to delivering meaningful advancements in early intervention orthopedic care, including osteoarthritis pain management, regenerative solutions, sports medicine, and Arthrosurface joint solutions.
Arthrosurface and Parcus have joined Anika.
Medical Professionals
We deliver a broad range of early intervention orthopedic solutions focused on joint preservation and restoration.
Patients
Learn more about our unique solutions for treating joint pain and arthritis, all designed and intended to restore active living.
Investors
We are committed to enhancing shareholder value through sustained profitable growth in early intervention orthopedics.
Our Vision
To be the global leader in joint preservation solutions that restore active living.
Our guiding principles are purpose driven and centered on customers and patients.
Join Anika
Everyone at Anika is passionate about helping people stay active and participate in the activities they love.
Let’s move forward together.
Since having my shoulders replaced, I have achieved 14 new world records, and I can honestly say that being treated with this alternative shoulder replacement has restored my life
Rudy Kadlub, Bilateral OVO with Inlay Glenoid Total Shoulder Arthroplasty Patient
View More Patient Success Stories